
If you have collected lichen material in the field, either for confirmation of identity, for adding to a reference collection (herbarium) the first thing you will want to do is take a quick look at your samples under a dissecting microscope and marvel at their beauty.
.jpg) Leave each packet or other container open to dry; damp lichens go mouldy and are of no use. Specimens should not be dried on hot radiators or in ovens as this may make them unsuitable for molecular studies by specialists.
Leave each packet or other container open to dry; damp lichens go mouldy and are of no use. Specimens should not be dried on hot radiators or in ovens as this may make them unsuitable for molecular studies by specialists.
Once dry, seal the packets in a plastic bag and place in a freezer for two days to kill off invertebrates, such as mites, which are likely to make a meal of your samples. (A domestic freezer is adequate for the purpose.) Freezing is also recommended if it is likely that molecular work may be undertaken – not necessarily by yourself, but perhaps by a referee to whom it is referred. Make sure that the packets don’t re-acquire moisture by condensation when you take them out of the freezer.
Microscopes
Two types of microscope are useful when examining lichens:
- A dissecting microscope with up to 40x (or more) magnification reveals the intricate world of lichens in their remarkable beauty – akin to seeing a landscape from far above and then suddenly being down at tree-top level – a breath-taking experience.
- A compound research microscope with x100, x400 and x1000 magnification and a calibrated eye-piece (graticule) for measuring anatomical details.
Using a microscope
When using a dissecting microscope, take a good look at specific structures such as soredia, isidia, pseudocyphellae, and pycnidia. It may be possible to identify the genus at this stage.
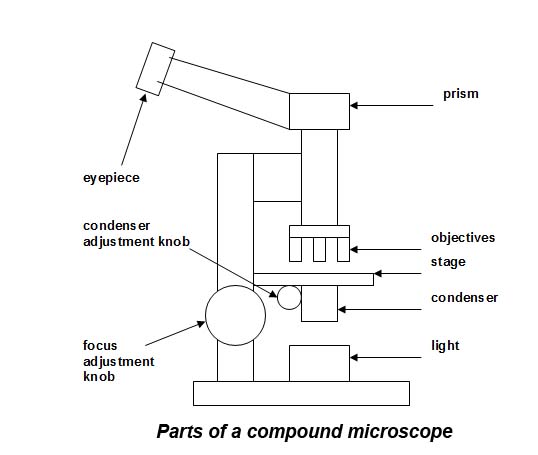
Useful items when using a compound microscope include:
- dissecting needles, a fine pair of tweezers, and a fine-pointed scalpel with a disposable blade;
- razor blades or scalpel blades for cutting sections: rigid single-sided razor blades can be obtained from microscope suppliers and scalpel blades from art material suppliers. Double-edged razor blades may be obtained from the high street shops but are not recommended as they are more likely to cause accidental cuts (and the blood may discolour the specimens!);
- slides and coverslips;
- an incident light source (usually included within the microscope casing).
Adjust a compound microscope to obtain maximum resolution as follows (see Figure above):
- place a slide with sample + coverslip on the stage and position it such that the sample covers only 1/3 of the field of view when seen through the x10 objective, and the sample is in focus;
- use the condenser adjustment knob to focus the light so that the image of the light source (bulb surface or filament) is clearly seen down the microscope, or place a scalpel tip on the light source and use this to focus;
- adjust slightly so that the image of the light (bulb surface or filament/scalpel tip) just disappears and even illumination is obtained;
- adjust the iris-diaphragm within the condenser to cut out stray light;
- the microscope is now ready for use.
Synchronising your graticule or ‘Is it really that small?’
 When comparing what you see with descriptions in The Lichens of Great Britain and Ireland (Smith et al 2009, 2011), there will be reference to the size of features in microns (µm). Such features are measured using a graticule placed in one of the eyepieces. A graticule is a measuring scale inserted in the eyepiece of the microscope which enables you to determine the size of microscopic features which are usually too small to be seen by the naked-eye: it must be calibrated using a stage-micrometer. (An expensive item which you will only need once; the Society has one which will be available for loan on field meetings and at other times by arrangement, and leaders of courses at Field Studies Council centres may well have one with them.) Once calibrated, and double-checked, the process need not be repeated unless accessories or different eyepieces or objectives are added to the microscope.
When comparing what you see with descriptions in The Lichens of Great Britain and Ireland (Smith et al 2009, 2011), there will be reference to the size of features in microns (µm). Such features are measured using a graticule placed in one of the eyepieces. A graticule is a measuring scale inserted in the eyepiece of the microscope which enables you to determine the size of microscopic features which are usually too small to be seen by the naked-eye: it must be calibrated using a stage-micrometer. (An expensive item which you will only need once; the Society has one which will be available for loan on field meetings and at other times by arrangement, and leaders of courses at Field Studies Council centres may well have one with them.) Once calibrated, and double-checked, the process need not be repeated unless accessories or different eyepieces or objectives are added to the microscope.
Spores, asci and paraphyses can now be measured using your graticule. Smaller spores and paraphyses are best measured using x100 oil-immersion objective; for medium and larger spores, the x40 objective may be satisfactory. When determining the size of a spore with the eye-piece graticule, take measurements from 10 spores (or as many as 30 spores to be statistically meaningful) selected at random and calculate an average. Take care that spores are lying flat, mature and well-formed. Measurements are usually given to the nearest micron or 0.5 micron to avoid a false impression of accuracy.
Preparing slides
Identifying the species requires detailed microscopic examination. This is often done by taking a VERY small amount and squashing it. Too much lichen on your slide and it will be almost impossible to spread out to see the relevant microscopic structures. More detailed examination may involve preparing at least 3 slides including:
- a thin vertical section to look at the spore-bearing structure (e.g. ascomata);
- a similar section for testing with chemicals;
- a section or small piece of the spore-bearing structure; squash, tap or tease this with a needle or pointed blade to see details of the ascus, ascospores, conidia etc.
To section a spore-producing body, place it on a slide and soak it in water for several minutes. Under the dissecting microscope, remove excess water with tissue-paper. Cut off and discard a segment from the outside and then cut a series of sections (as thin as possible) with a razor or scalpel blade. Pick up the sections with a minute drop of water using a dissecting needle or razor-blade and place in the drop of water on the slide. Some lichens can be sectioned more effectively using dry material; you will need to experiment to find out what works best for you. Section-cutting takes practice; the most common mistake beginners make is to try (usually unsuccessfully) to view lichen structures in a section that is too thick.
To prepare the asci for staining, remove most of the water with tissue and replace it with 5% KOH (potassium hydroxide). Leave a few minutes and then remove the KOH with tissue and replace with acidified water (50 ml water with a few drops of strong acid) to neutralise the residual KOH. Blot and repeat to rinse the KOH away. Add a drop of Lugol’s iodine, and leave for a few moments to stain the asci and then add a coverslip.
There are different mountants that can be used to preserve slides, as those prepared in water or KOH, will soon dry out. The most commonly used mountant for semi-permanent preparations is lactic acid with cotton blue (which can be purchased made-up from some laboratory suppliers). To use this, place a section or piece to be squashed directly in the smallest drop of the mountant that will allow a coverslip to lie flat on it, warm over a spirit lamp or similar heat-source taking care it does not boil and splutter the specimen away, allow to cool, blot away any excess mountant, and seal with nail-varnish or a proprietary slide sealant. Warming is necessary as otherwise dimensions of microscopic features such as spores may be artefactually reduced in size.
Note that if a slide is particularly fine, even if made with a mountant, it will soon dry out; a spot of nail varnish on one side of the coverslip will keep it in place so that it can be wrapped in tissue and kept with the specimen. Such temporary slides can then be re-hydrated with a drop of water and used for comparisons; if enclosed with specimens sent to a referee they can also save time and may speed up a response!
John Skinner has produced a useful guide to preparing sections which can be downloaded here - Sectioning: some ideas that might help. This was published in the Bulletin in 2016.
Sections, Staining, Ascus tips and chemicals
Read Mark Powell's advice on microscope sections, staining, ascus tips and chemicals.
Recording what you see
Sketch or photograph what you see and record measurements taken. Keep the notes with the specimen so that new preparations do not have to be made to compare it with others later. This is essential if sending the specimen off to a referee, as it saves a lot of time and makes a prompt response more likely.
Safety
Dispose of razor blades, scalpel blades, needles, broken slides and coverslips in a robust rigid pot.